
Climate
A digital platform to recognize informal waste recovery in Colombia
Impact
News and Insights
HemoClear BV in partnership with Nigeria's National Blood Service Agency (NBSA) seeks to address the transfusion blood scarcity in Nigeria through an innovative technology. The purpose of this project is to understand the optimal method of an autologous blood transfusion service implementation in low- and middle-income countries healthcare setting. The project will target primarily women giving birth and undergoing gynecological procedures in two hospitals spread over two regions in the country.
Project ported by:


Many countries especially those with low and middle incomes face challenges in obtaining safe blood and its transfusion products, even though they are essential for critical medical procedures like cardiac surgeries, organ transplants, cancer treatments, childbirth and trauma care. This scarcity poses constraints on healthcare systems and affects their ability to deliver optimal healthcare services of high quality (Oreh and Amedu, 2023).
In Nigeria, the situation is critical. Each year, 80,000 women die during childbirth due to insufficient access to safe blood. According to the World Health Organization (WHO) (2023), only 27% of the demand for blood in the country is met. This scarcity leads to an increased risk of transfusion-related infections from inadequately screened paid donors, particularly impacting maternal health, while also contributing to 10% of all new HIV infections.

HemoClear BV has developed a microfiltration technology to isolate blood cells from a liquid component (Amenge et al., 2022). This blood separation device is considered to make the blood collection and blood cleaning process easier and faster, to reduce the cost of blood transfusion and to increase the patient's recovery (Carroll C, Young F., 2021). Delivered in a kit including all the needed materials, it could be used by any medical staff after a short instruction.
HemoClear BV in partnership with the Nigeria's National Blood Service Agency (NBSA) proposes to spread the use of this autologous blood transfusion system, already tested in the United Kingdom, in Nigeria's hospitals. FID’s funding will allow to test the feasibility of transposing this solution to Nigeria initially targeting two hospitals, the Calabar University Teaching hospital and the Abuja Teaching hospital.
HemoClear’s team also aims to share study data for the World Health Organization (WHO) approval and inclusion of the innovation in the WHO open access electronic database of Medical Devices, Priority Medical Devices Information System (MeDevIS).
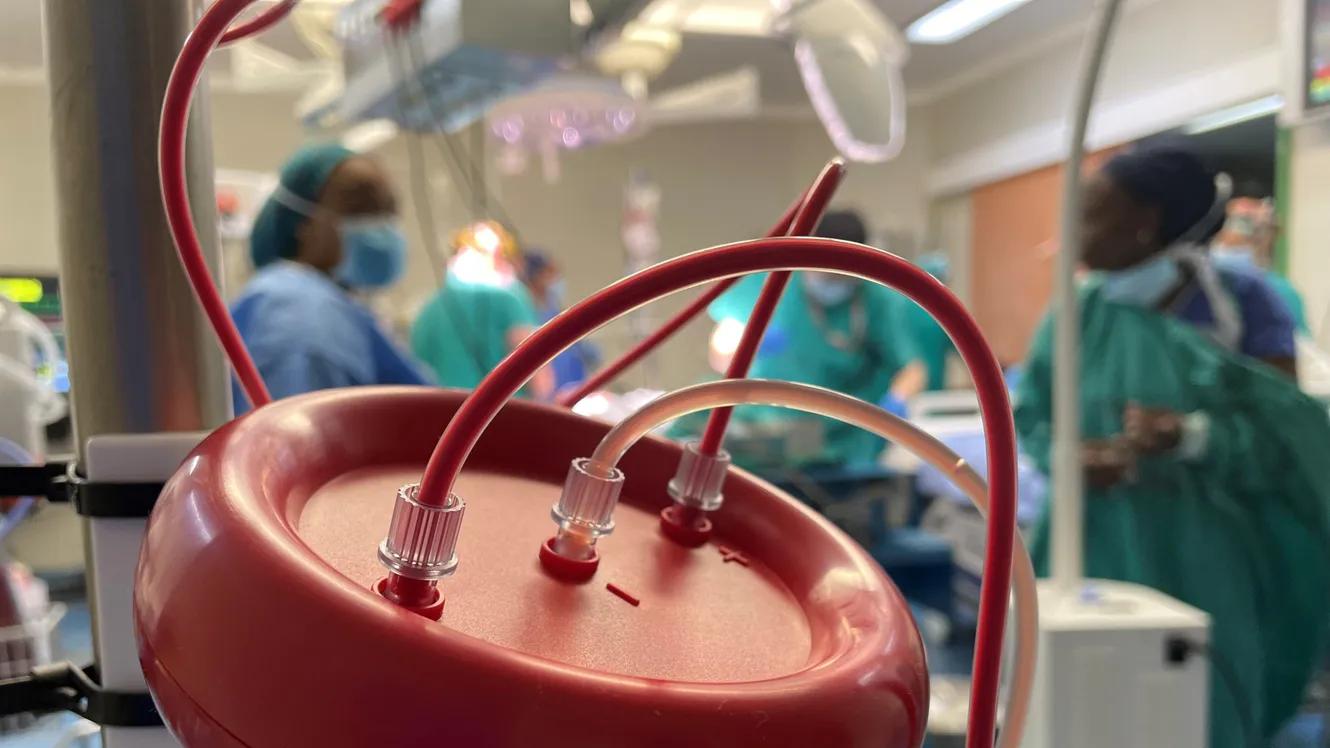
The objective of the project is to reduce the consequences of blood shortages and unsafe transfusions. The pilot will test the project in the context of a low- and middle-income country, focusing on mothers during childbirth.
It will allow to produce learnings on the optimal training method for adoption of HemoClear’s device, the practical and clinical requirements for its implementation and adoption, and the feasibility of a national autologous blood transfusion service provided by the Nigeria's National Blood Service Agency (NBSA).
Projects
Projects funded by FID